The bioequivalence can play a vital role in the drug industry that is used to balance the chemical compounds. The dosage level of every drug is the most mandatory thing to be considered or else it may have a chance to create wide ranges of health problems to the patients. The bioequivalence studycan take various kinds of factors into the considerations such as body weight, sex, age, physical, and mental health condition for providing proper medicine to the patients as per their health condition.
There are numerous amounts of advanced technologies have been established in the drug industry to enhance the benefits of consumers. While dealing with the bioavailability study, the professionals need to consider wide ranges of factors that are mentioned below:
Focus On Crossover Design
When comes to the crossover design, the test drug needs to be compared with the standard reference. Here, the professionals need to make sure whether the drugs are perfectly suitable for the 18-55 years age-grouped people. If any of the misunderstandings can happen in this section, then it may have a chance to produce huge health impacts on the consumers. In this phase, there are two types of procedures have been carried out to identify the washout period of every drug.
Washout Period Of The Drugs
The term washout period is nothing but the time lying between the treatment periods. The main objective of this phase is to eliminate the administered dosage present in each and every drug. The number of washout period carried out in the process can strongly depends upon the type of crossover design used in the drug. Furthermore, the number of formulations evaluated using Latin square design can be clearly understood via the process chemistry. Some special types of equipment have been introduced in this field to simplify the burden of the professionals.
Parallel Design In Bioavailability Studies
When comes to the parallel design, the two different formulations are given to the groups of volunteers. These two formulations are administered randomly to avoid bias. However, the intersubject variations cannot be identified exactly in this process. It has been proved effectively via the bioequivalence study and the process shows greater variation between all kinds of the formulation.
This kind of process is carried out to eliminate the excess dosage level present in the drugs. After the completion of the entire drug work, the bioequivalence process is conducted to balance all the chemical compounds used in it.
Why Bioequivalence In Drug Industry?
The main objective of this process is to find out the dosage level of the drug prepared in the industry. The health condition of the patient can differ from one another and so the level of the dosage should be at a medium level. Both the single-dose and the multi-dose standard is involved in this process chemistry for evaluating the parameters of each drug.
Thus, these are all the significant factors have been carried out in the drug industry to evaluate the chemical compounds included in it. hope, the above-written article will help you to understand the process of drug evaluation.
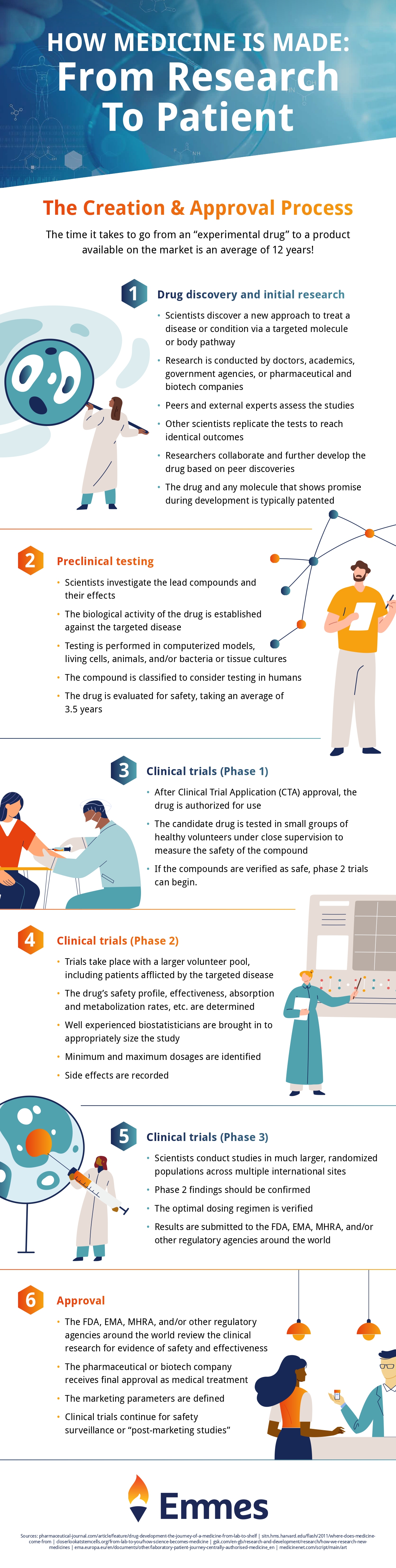
Drug evaluation is just one stage in the medicine making process. Check out the infographic below to see how your medicine is made, from start to finish!
Infographic provided by The Emmes Company, a clinical data management organization